News
| Japan Cytometry Society, Kobe | Lecture | M.Inoue | |
| National Cancer Center, Tokyo | Presentation | M.Inoue | |
| Life Science Week, Tokyo | Presentation | M.Inoue | |
| Japanese Society for Regenerative Medicine, Sendai | Presentation | M.Inoue | |
| Kumamoto University, Kumamoto | Presentation | M.Inoue | |
| Cell Assay Symposium, Tokyo | Presentation | M.Inoue | |
| Japanese society for Radiation Oncology, Kyoto | Presentation | M.Inoue | |
| Epithelial Biology Center Seminar, Vanderbilt Univ., Nashville | Lecture | M.Inoue | |
| National Institute of Biomedical Innovation, Health and Nutrition, Osaka | Lecture | M.Inoue | |
| Japanese Cancer Association, Yokohama | Presentation | M.Inoue | |
| Charles River Short Course, Yokohama | Lecture | M.Inoue | |
| Nagahama Institute of Bio-Science and Technology, Shiga | Lecture | M.Inoue | |
| Okinawa Institute of Advanced Sciences, Uruma | Lecture | M.Inoue | |
| Safety Evaluation Forum, Suwa | Lecture | M.Inoue | |
| Korean Cancer Association, Seoul | Presentation | M.Inoue | |
| Inha University, Incheon | Presentation | M.Inoue | |
| Japan Society for Cell Biology, Kyoto | Presentation | M.Inoue |
Research
#1 Primary culture of cancer cells: CTOS method
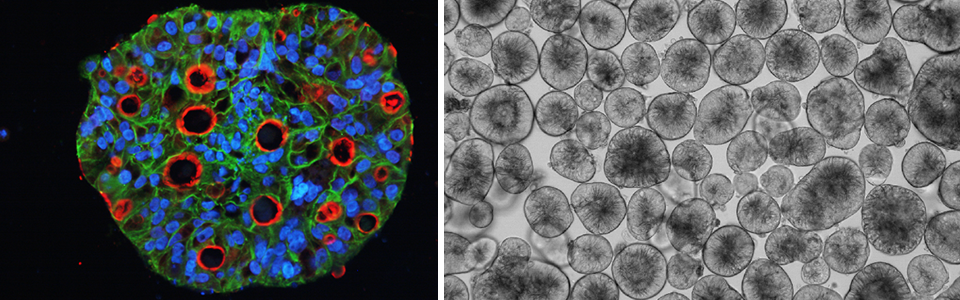
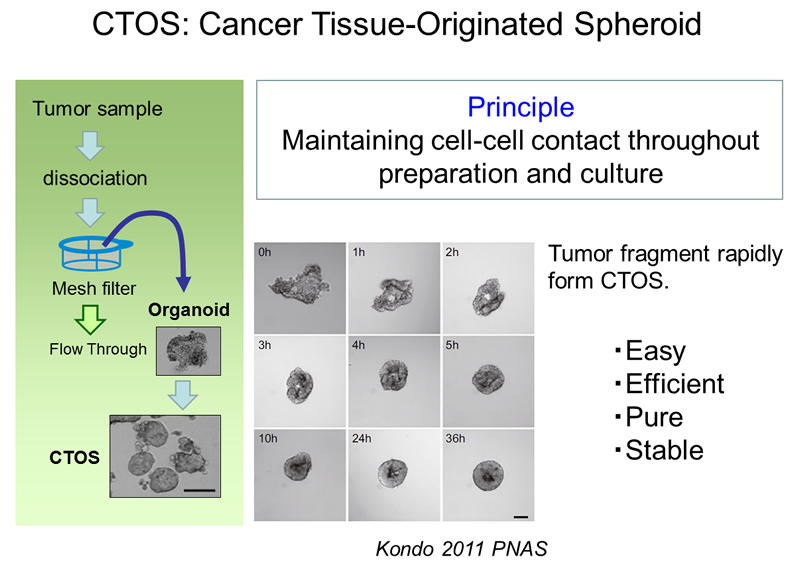
CTOS (cancer tissue-originated spheroid) method was developed in our laboratory (1). The method is for preparation and culture of cancer cells from tumors, based on the principle of maintaining cell-cell contact throughout preparation and culture. Dissociating tumors into single cells has long been a gold standard in primary culture of cancer cells. However most of the cancer cells in solid tumors exist maintaining cell-cell interaction with other tumor cells.
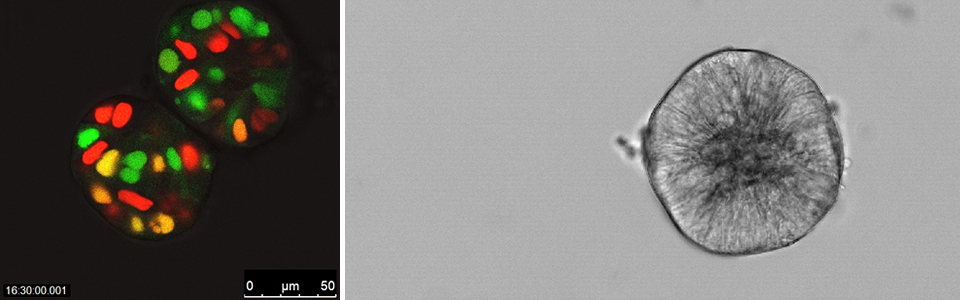
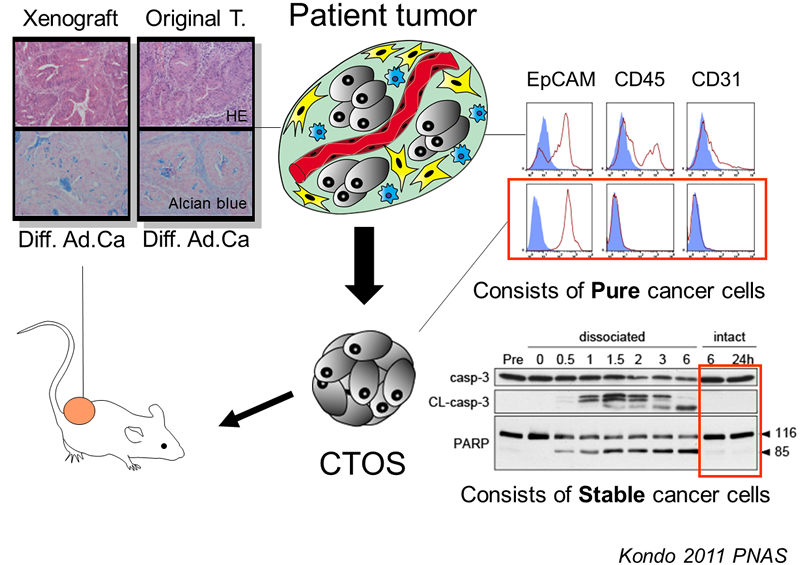
The idea of CTOS method came up with the finding that cancer cells drastically die when they are dissociated into single cells. Therefore we partially dissociate the tumors, filter, and collect the cell clusters upon the filter, discarding the single cells in the flow-through fraction (Figure 1). Another critical finding in the development of CTOS method was that the cell clusters autonomously form spheroids within a short period, few hours. We call these spheroids, which are formed from undissociated clusters, as CTOS. In contrast to the cells dissociated into single-cell, the cells within a CTOS are quite stable (Figure 2), so that cancer cells can be prepared quite efficiently from patient tumors. In addition to the high feasibility, CTOS consists of pure cancer cells. Moreover, CTOS preserves characteristics of original patient tumors in vitro as well as in CTOS-derived xenograft tumors. Thus CTOS provides the opportunity for the study that are clinically relevant.
1. Kondo J, Endo H, Okuyama H, Ishikawa O, Iishi H, Tsujii M, Ohue M, Inoue M. Retaining cell-cell contact enables preparation and culture of spheroids composed of pure primary cancer cells from colorectal cancer. Proc Natl Acad Sci U S A. 2011;108(15):6235-40.
#2 Hypoxia response of cancer cells
Tumor microenvironment is hypoxic. Cancer cells respond to hypoxia in various ways, which can be classified into three categories: 1) adaptation to hypoxia, 2) renovation of microenvironment, and 3) escape from hypoxia. All of the responses relate to malignancy. Cancer cells adapt to hypoxia by altering metabolism or inducing anti-apoptotic pathway. Hypoxia is one of the strongest inducers of angiogenesis, which resolve the hypoxic microenvironment. Cancer cells in hypoxia escape from deteriorated microenvironment by activating invasion or metastasis. Among these responses, we have been focusing on the adaptation to hypoxia, especially on tumor cell dormancy.
HIF-1a, hypoxia inducible factor-1a, is a transcription factor which plays major roles in the transcriptional regulation in acute hypoxia response. One of the drastic responses is the increase of glucose utilization by activating glycolytic pathway and suppressing pyruvate influx into mitochondria, which are all regulated by the downstream genes of HIF-1a. The paradox in tumor hypoxia is that glucose and other nutrients are also poorly supplied in the region nearby the diffusion limit of oxygen. It is catastrophic for the cancer cells to increase consumption of energy source when the supply is decreased. Therefore, there must be another adaptation mechanism than HIF-dependent responses to acute hypoxia. We are studying tumor cell dormancy as an alternative but critical adaptation mechanism in hypoxia (2).
2. Endo H, Okami J, Okuyama H, Nishizawa Y, Imamura F, Inoue M. The induction of MIG6 under hypoxic conditions is critical for dormancy in primary cultured lung cancer cells with activating EGFR mutations. Oncogene. 2016.
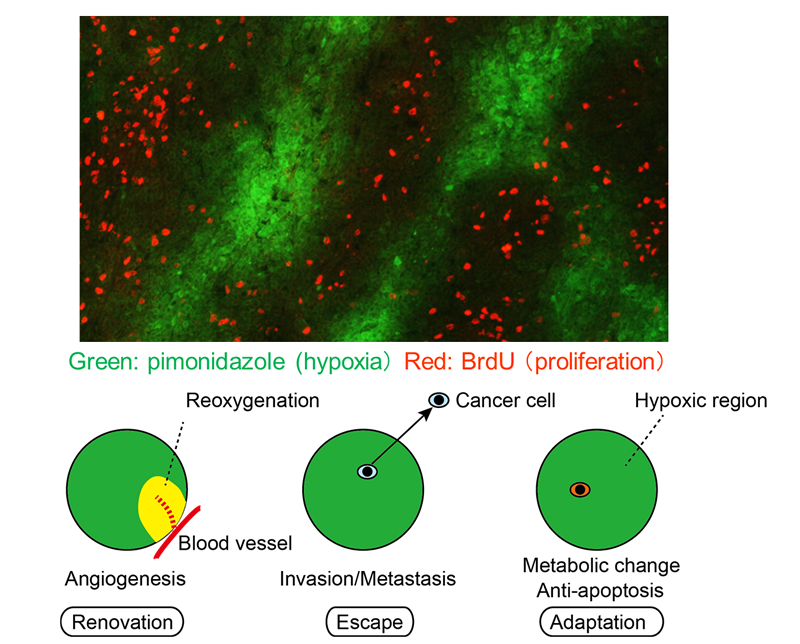
Figure: Hypoxia adaptation
Paper
2017
- Tashiro T, Okuyama H, Endo H, Kawada K, Ashida Y, Ohue M, Sakai Y, Inoue M. In vivo and ex vivo cetuximab sensitivity assay using three-dimensional primary culture system to stratify KRAS mutant colorectal cancer. PLoS One. 2017
2016
- Arai M, Kawachi T, Kotoku N, Nakata C, Kamada H, Tsunoda S, Tsutsumi Y, Endo H, Inoue M, Sato H, Kobayashi M. Furospinosulin-1, Marine Spongean Furanosesterterpene, Suppresses the Growth of Hypoxia-Adapted Cancer Cells by Binding to Transcriptional Regulators p54(nrb) and LEDGF/p75. Chembiochem. 2016;17(2):181-9.
- Hayashi N, Kataoka H, Yano S, Kikuchi J-I, Tanaka M, Nishie H, Kinoshita Y, Hatano M, Nomoto A, Ogawa A, Inoue M, Mizoshita T, Shimura T, Mori Y, Kubota E, Tanida S, Joh T. Anticancer Effects of a New Aminosugar-conjugated Platinum Complex Agent Against Cisplatin-resistant Gastric Cancer. Anticancer Research. 2016;36(11):6005-10.
- Hiraide T, Ikegami K, Sakaguchi T, Morita Y, Hayasaka T, Masaki N, Waki M, Sugiyama E, Shinriki S, Takeda M, Shibasaki Y, Miyazaki S, Kikuchi H, Okuyama H, Inoue M, Setou M, Konno H. Accumulation of arachidonic acid-containing phosphatidylinositol at the outer edge of colorectal cancer. Sci Rep. 2016;6:29935.
- Kimura H, Fumoto K, Shojima K, Nojima S, Osugi Y, Tomihara H, Eguchi H, Shintani Y, Endo H, Inoue M, Doki Y, Okumura M, Morii E, Kikuchi A. CKAP4 is a Dickkopf1 receptor and is involved in tumor progression. J Clin Invest. 2016;126(7):2689-705.
- Kiyohara Y, Yoshino K, Kubota S, Okuyama H, Endo H, Ueda Y, Kimura T, Kimura T, Kamiura S, Inoue M. Drug screening and grouping by sensitivity with a panel of primary cultured cancer spheroids derived from endometrial cancer. Cancer Sci. 2016;107(4):452-60.
- Narumi A, Tsuji T, Shinohara K, Yamazaki H, Kikuchi M, Kawaguchi S, Mae T, Ikeda A, Sakai Y, Kataoka H, Inoue M, Nomoto A, Kikuchi J, Yano S. Maltotriose-conjugation to a fluorinated chlorin derivative generating a PDT photosensitizer with improved water-solubility. Org Biomol Chem. 2016;14(14):3608-13.
- Okuyama H, Kondo J, Sato Y, Endo H, Nakajima A, Piulats JM, Tomita Y, Fujiwara T, Itoh Y, Mizoguchi A, Ohue M, Inoue M. Dynamic Change of Polarity in Primary Cultured Spheroids of Human Colorectal Adenocarcinoma and Its Role in Metastasis. Am J Pathol. 2016;186(4):899-911.
- Sato Y, Tateno H, Adachi J, Okuyama H, Endo H, Tomonaga T, Inoue M. Generation of a monoclonal antibody recognizing the CEACAM glycan structure and inhibiting adhesion using cancer tissue-originated spheroid as an antigen. Sci Rep. 2016;6:24823.
- Tanaka M, Kataoka H, Yano S, Sawada T, Akashi H, Inoue M, Suzuki S, Inagaki Y, Hayashi N, Nishie H, Shimura T, Mizoshita T, Mori Y, Kubota E, Tanida S, Takahashi S, Joh T. Immunogenic cell death due to a new photodynamic therapy (PDT) with glycoconjugated chlorin (G-chlorin). Oncotarget. 2016.
Members
Principal Investigator
Masahiro INOUE(detail)
Senior Researchers
Hiroko ENDO
Jumpei KONDO
Postdoctoral fellows
Yumi SATO
Roberto COPPO
Graduate students
Takeshi HAGIHARA (Kyoto Univ.)
Yu ITO (Osaka Univ.)
Mie TANAKA (Osaka Univ.)
Kumiko TATSUMI (Kyoto Univ.)
Technical Staffs
Noriko KANTO
Tomoya EKAWA
Arisa TSUDA
Secretaries
Toshiko YASUDA
Masami IZUTSU
Location
Location
Department of Biochemistry
Osaka International Cancer Institute
3-1-69 Otemae, Chuo-ku, Osaka, Japan